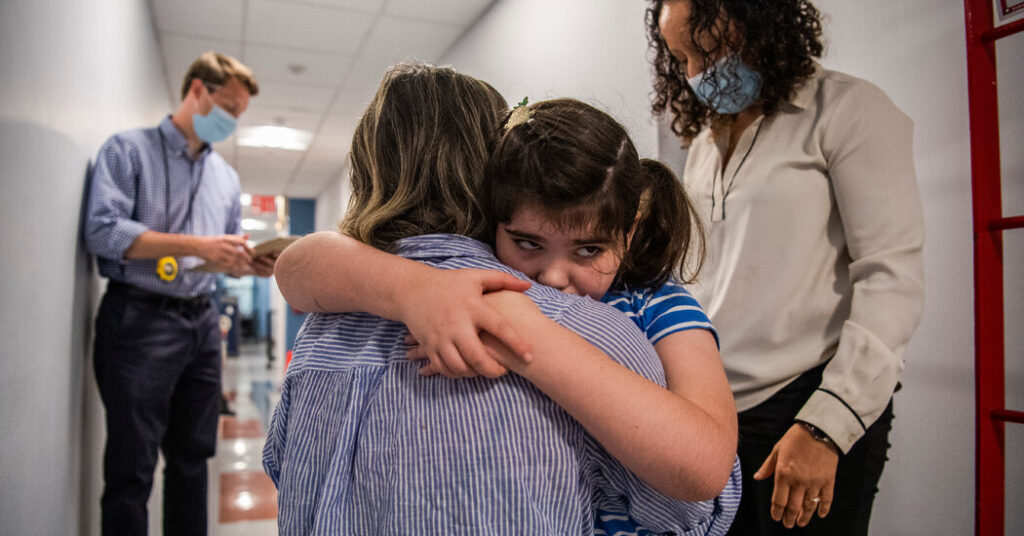Susannah Rosen, 8, spent much of her childhood in hospitals in New York City as doctors documented the gradual loss of her ability to stand, walk and see.
But on a visit this October, her parents thought for the first time that she might leave the hospital better off than before. That’s when surgeons infused a drug into her spine to fix the ultrarare genetic glitch that had vexed her nervous system since infancy.
“Every other time we go into the hospital, it’s because something terrible has happened,” said Susannah’s father, Luke Rosen. “This time, there was hope for something that will heal her.”
Susannah was the first person to receive a drug designed to treat KIF1A-associated neurological disorder, or KAND, a progressive disease caused by genetic mutations that affect just 400 people in the world. In doing so, the young girl and her parents have found themselves on the edge of personalized medicine.
Since the technology for such bespoke genetic drugs debuted in 2018, about two dozen patients have received the infusions — costing as much as $2 million per patient — to treat a range of neurological syndromes. But hundreds of millions of others, mostly children, live with rare genetic diseases and have no treatment options.
Susannah’s drug, almost two years in the making, was paid for by a nonprofit organization, n-Lorem, that aims to do the same for at least 1,000 patients over the next decade. By raising funds and negotiating discounts and in-kind donations from biotechnology companies to make its drugs, n-Lorem’s founder believes, the organization can fulfill its mission to “leave no child behind.”
But other rare-disease experts doubt that a funding model based on donations will ever be large or sustainable enough to help millions of patients. They are searching for other ways to accelerate the technology’s development, which includes seeking help from for-profit businesses.
Groups developing therapies for these diseases must also grapple with how to share valuable — and rare — data. N-Lorem has been criticized for not pledging to share information about its patients and methods swiftly and transparently, an issue that became more urgent after a girl died last year from complications in a clinical trial.
“These are really complex questions that this field has opened up,” said Issi Rozen, a venture partner at GV, formerly known as Google Ventures, a firm that has invested in the field. “The worst thing I can imagine is those technologies exist that can treat the kids, but there’s no framework for doing that.”
Cutting Costs
Susannah’s parents first noticed something wrong with their daughter when she was a baby and couldn’t kick her legs in the bathtub. As a toddler, she used leg braces to stand and walk and would fall suddenly. When Susannah was 2, doctors discovered that she had seizures while she slept.
In 2016, her parents learned that she carried a glitch in a gene, called KIF1A, which caused KAND. The untreatable disease, her doctor said, would cause developmental delays, vision loss, seizures and physical disabilities that would worsen with time.
“What can we do to fix that?” Mr. Rosen recalled asking Wendy Chung, Susannah’s doctor and a pediatrician and geneticist at Columbia University.
Dr. Chung advised them to find other patients. Mr. Rosen and his wife, Sally Jackson, started a foundation in 2017, found about 400 other patients, raised $2 million for research and began lobbying scientists to develop treatments.
One of the companies Mr. Rosen called was Ionis Pharmaceuticals, based in Carlsbad, Calif. Ionis used snippets of genetic material — known as antisense technology — to make drugs for diseases that are somewhat rare, affecting tens of thousands of people in the United States, but much more common than KAND.
The next year, Dr. Timothy Yu, a neurologist and genetic researcher at Boston Children’s Hospital, announced that over the course of just 10 months he had developed a customized antisense drug for an 8-year-old girl named Mila Makovec. The drug, named milasen after its patient, treated Mila’s rare neurological condition, Batten disease.
The founder of Ionis, Dr. Stanley Crooke, also wanted to treat extremely rare diseases, but he believed companies could not profit from drugs used by fewer than 30 people. So in 2020, he and his wife, Rosanne Crooke, started the n-Lorem foundation with two founding partners, Ionis and Biogen, a biotechnology company in Cambridge, Mass. Since then, n-Lorem has raised $40 million to make such drugs.
Ionis and other companies agreed to provide discounted or free equipment and services, including drug manufacturing and safety testing. In turn, n-Lorem would provide the infusions to patients for free, indefinitely.
“Developing, manufacturing and then giving it away for life for free is an amazing concept, for the most desperate, most underserved patient population we know of,” Dr. Crooke said.
N-Lorem has so far enrolled more than 80 patients, including Susannah, for this lifetime treatment plan, and its leaders hope to treat many more in the coming years. Dr. Crooke said that drug manufacturing discounts and efficiencies cut the cost of making each individualized therapy by as much as 40 percent. Dr. Yu at Boston Children’s needed $2 million to make the drug for Mila, for example. But n-Lorem has cut that cost to an average of $800,000 per patient, Dr. Crooke said.
It took 17 months for scientists at n-Lorem to create a drug to shut down Susannah’s specific glitch in the KIF1A gene, which is not shared by any other patient in the country.
While waiting, Susannah grew sicker. She had broken several bones from falls and used a wheelchair much of the time. Her vision gradually faded. Whenever Mr. Rosen traveled, he worried that his daughter wouldn’t be able to see his face when he got home.
The new drug would not cure Susannah, her parents knew, but they hoped it would alleviate her seizures and difficulties with motor control. Maybe by Christmas, they thought, she would be able to walk to her brother and hug him.
On Oct. 10, Dr. Jennifer Bain, a pediatric neurologist at NewYork-Presbyterian Morgan Stanley Children’s Hospital in Manhattan, injected Susannah’s drug into her spine.
The next morning, Susannah woke up smiling. Her parents wondered if the drug was already working, though they knew that was unlikely.
Mr. Rosen and Ms. Jackson logged her seizures and falls daily. Neurologists planned to examine Susannah’s mental abilities, brain activity and movement skills every few weeks.
Susannah was the first patient to receive an n-Lorem drug, followed by two adult patients in October and November.
Growing Pains
Some rare disease experts are skeptical that one nonprofit organization will be able to serve every patient who needs help.
Some are searching instead for a viable business model that could bring millions or billions of dollars of investor funding. The money is needed to hasten the competition necessary to drive down costs, prove the medicines work and convince insurers to pay for them.
In 2021, Julia Vitarello, the mother of Mila, co-founded EveryONE Medicines, a for-profit company in Boston that is exploring how to make customized genetic drugs sustainably.
And Jeff Milton, a former Ionis scientist, hopes to develop rare-disease-drugs that target biological systems that are also affected in more common diseases. That could coax investors to invest in his start-up, La Jolla Labs, to develop drugs that could treat both rare and common diseases, he said.
Both are also focused on how various outfits can share data.
Ms. Vitarello also founded a nonprofit organization with Dr. Yu, called the N=1 Collaborative, that aims to make personalized medicines more accessible. Its 311 members, including parents, patients, investors and scientists from academia, companies and other institutions, have pledged to share information with each other.
“We’re talking about dying children,” Ms. Vitarello said. Mila died at age 10, three years after receiving the first dose of her customized drug. “Companies, academic institutions and foundations should all have a mandate to share data so we can learn what works, because it is unethical not to.”
These tensions have increased since the recent death of a child who received a customized antisense drug.
On Oct. 23, Dr. Yu reported at a scientific meeting that two of his patients developed a buildup of fluid in the brain, called hydrocephalus, after receiving a drug for a severe form of epilepsy, and one died. He and other scientists are studying whether other antisense drugs might cause the same problem.
Dr. Yu announced the results before publishing them in a peer-reviewed journal article, he said, partly to highlight the importance of sharing data through channels such as the N=1 Collaborative.
Dr. Crooke said that n-Lorem would not contribute its data to the N=1 Collaborative database. The organization has presented data at scientific conferences, he said, and will publish data on its patients in peer-reviewed journals. It will also alert the Food and Drug Administration if a death or serious adverse event occurs.
He said he did not think n-Lorem’s data should be compared with others’ because the n-Lorem team had more expertise in making antisense drugs, also known as ASOs. “We’re not going to mix data from our optimized ASOs with data from ASOs that are not optimized,” Dr. Crooke said.
But Dr. Yu said that Dr. Crooke’s assertion that n-Lorem’s drugs were superior was “unjustified and easily refutable.” For instance, a clinical trial using an antisense drug licensed from Ionis caused hydrocephalus in patients with Huntington’s disease who received the highest doses.
About two dozen patients have received customized antisense drugs since Dr. Yu’s announcement about Mila in 2018. His team has treated four other patients, including the two children who developed hydrocephalus.
N-Lorem is racing to make drugs for the patients it has enrolled. The organization hopes to add 100 to 150 patients to its list per year, reaching about 1,000 patients within a decade.
Dr. Yu and others say that if they can show that the drugs save lives, investors might step in.
“Ultimately, before this explodes into treating dozens of families per year, they’re going to have to show that it works,” said David Corey, a biochemist at the University of Texas Southwestern Medical Center at Dallas, who is not involved in the antisense field.
On Nov. 9, Susannah returned to the hospital for the second dose of her drug. After the procedure, she nestled with dolls in bed and chatted with her parents, at times looking them straight in the eye. This was unusual; her vision problems usually forced her to use her peripheral vision.
Susannah unscrewed the lid of a doll’s pink plastic sippy cup and held the cup out to Mr. Rosen.
“Daddy, can you fill this up with water?” she asked.
Mr. Rosen obliged. He thought that her speech had recently improved and was impressed with her ability to focus her gaze more clearly.
Less than a month later, Susannah surprised her parents by standing up on her own for the first time in two years. After pulling herself up from the living room carpet, Susannah, face flushed with exertion, stood tall and high-fived Ms. Jackson.
Susannah received her third dose of the drug on Dec. 7. With four months to go in the trial, Mr. Rosen and Ms. Jackson felt cautiously optimistic.
“It’s just not easy to be Susannah because there’s no road map,” Mr. Rosen said. “She’s creating it.”
You may also like
-
Bills co-owner Kim Pegula escorted on field in latest sign of her recovery from cardiac arrest
-
Abortion-rights extremists to apologize, pay restitution for attacks on pro-life facilities
-
Biden's virus symptoms are gone, says his doctor
-
Jon Stewart pushes VA to help veterans sickened after post-9/11 exposure to uranium
-
Study links marijuana use in pregnant women to preterm birth risks